Startups
German BioTech Exciva secures €51 million funding for innovative Alzheimer’s drug testing in patients
Exciva, a biopharmaceutical company based in Heidelberg, has successfully closed a Series B funding round, raising €51 million ($59 million). The round was co-led by Gimv and EQT Life Sciences, with participation from new investors Fountain Healthcare Partners, LifeArc Ventures, Carma Fund, and Modi Ventures, as well as existing investors Andera Partners and LBBW.
François Conquet, CEO of Exciva, expressed delight in attracting funding from both existing and new investors, highlighting the promising potential of their product. He emphasized the significance of positive results from the phase 2 trial in advancing symptomatic treatment options for patients with Alzheimer’s disease.
In the European landscape of Alzheimer’s and neurodegeneration funding, Exciva’s Series B joins other notable financings, including Augustine Therapeutics, Aerska, TRIMTECH Therapeutics, and Punto Health. These rounds collectively reflect a substantial investment of approximately €126 million in therapeutics and solutions related to Alzheimer’s and dementia.
Exciva’s financing stands out as one of the larger late-stage rounds in the segment, showcasing continued investment in disease-modifying approaches and treatments for neuropsychiatric symptoms associated with Alzheimer’s disease.
Andreas Jurgeit, a partner at Gimv, praised Exciva’s differentiated therapy for agitation in Alzheimer’s disease, highlighting its potential to address significant gaps in current treatment options.
Founded in 2016 by Drs. Anton Bespalov, Hans Moebius, and Rao Vepachedu, Exciva focuses on addressing neuropsychiatric symptoms in Alzheimer’s disease dementia and other brain disorders. The company aims to provide innovative solutions for the challenging aspects of dementia care.
The global prevalence of Alzheimer’s disease is on the rise, with behavioral and psychological symptoms posing significant challenges for patients and caregivers. Exciva’s research and development efforts aim to address these issues and improve the quality of life for individuals affected by the disease.
The latest funding will support a phase 2 study evaluating the therapeutic potential of Deraphan in treating agitation in Alzheimer’s patients. Deraphan, a combination of two CNS-active compounds, has shown promise in offering improved efficacy and safety compared to existing therapies. The clinical trial will be conducted in multiple countries, including Europe, the UK, the US, and Canada.
Exciva’s board of directors, post-Series B, includes representatives from Andera Partners, EQT Life Sciences, Gimv, Fountain Healthcare, an independent board member, and CEO François Conquet.
Overall, Exciva’s innovative approach to addressing neuropsychiatric symptoms in Alzheimer’s disease demonstrates a commitment to advancing patient care in this challenging field. The company’s dedication to scientific excellence and patient-centric focus positions it as a key player in the development of novel therapies for Alzheimer’s and related conditions.
-

 Facebook5 months ago
Facebook5 months agoEU Takes Action Against Instagram and Facebook for Violating Illegal Content Rules
-

 Facebook5 months ago
Facebook5 months agoWarning: Facebook Creators Face Monetization Loss for Stealing and Reposting Videos
-

 Facebook5 months ago
Facebook5 months agoFacebook Compliance: ICE-tracking Page Removed After US Government Intervention
-

 Facebook4 months ago
Facebook4 months agoFacebook’s New Look: A Blend of Instagram’s Style
-

 Facebook4 months ago
Facebook4 months agoFacebook and Instagram to Reduce Personalized Ads for European Users
-
 Facebook5 months ago
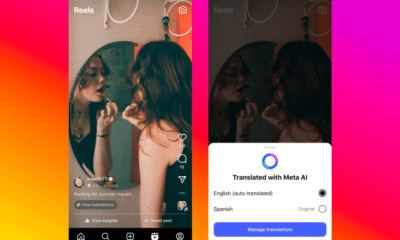
Facebook5 months agoInstaDub: Meta’s AI Translation Tool for Instagram Videos
-
 Facebook4 months ago
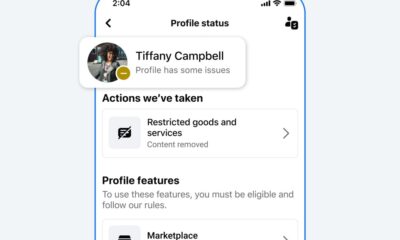
Facebook4 months agoReclaim Your Account: Facebook and Instagram Launch New Hub for Account Recovery
-

 Apple5 months ago
Apple5 months agoMeta discontinues Messenger apps for Windows and macOS






























