Mobile Tech
Apple Studio Display XDR Receives FDA Approval for Medical Applications
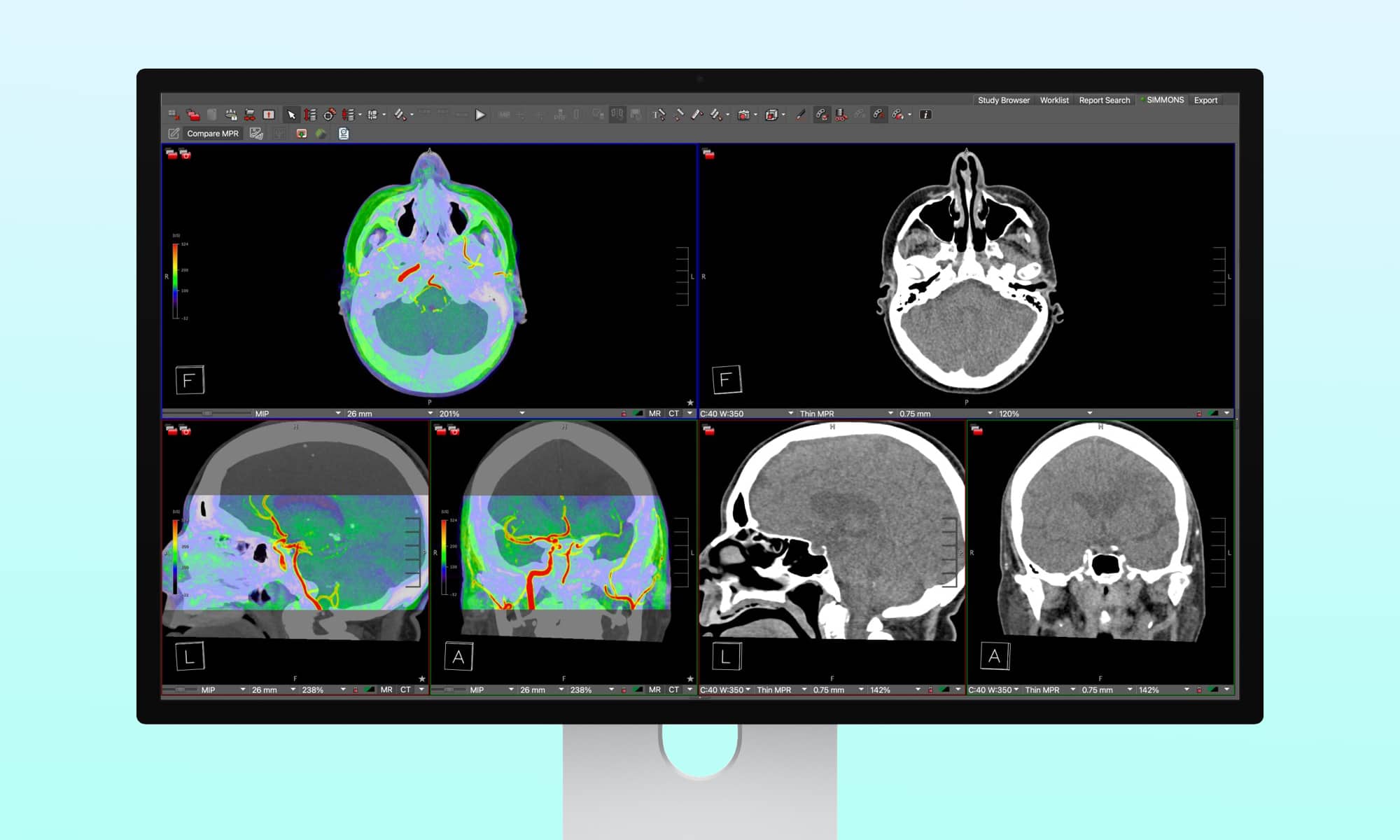
Apple’s latest Studio Display XDR has recently received approval from the US Food and Drug Administration (FDA) for use in diagnostic medical imaging, marking a significant milestone for the device. This development not only highlights the display’s suitability for professional photo and video editing tasks but also underscores its precision and reliability in critical healthcare applications.
The capability to support medical imaging is one of the 16 Reference Modes offered on the Apple Studio Display XDR. When the display was introduced in early March, Apple included “Medical Imaging (DICOM-350 nits)” and “Medical Imaging (DICOM-600 nits)” among the available modes. However, it was specified that these modes were intended for medical professionals and not for mammography.
Furthermore, the utilization of a “Medical Imaging Calibrator” tool in macOS was deemed necessary for these medical imaging features, which was not initially accessible. Apple elaborated on the medical imaging functionalities in its Studio Display XDR Technology Overview, indicating an expected availability by April 2026.
The Studio Display XDR now supports medical imaging for diagnostic radiology, provided that the new medical imaging presets and Medical Imaging Calibrator within macOS are utilized. These features are designed for trained medical professionals in general radiology, excluding mammography. The display incorporates two distinct medical imaging reference modes to cater to the preferences of radiologists.
Obtaining FDA clearance for medical diagnostic use is crucial, with the focus primarily on the “Medical Imaging Calibration Feature (MICF).” The FDA granted approval on April 1, approximately four months after the feature was initially submitted on November 17, 2025, highlighting the extensive planning required by Apple for such functionalities.
Greg Joswiak, Apple’s Senior VP of Worldwide Product Marketing, confirmed the availability of the MIC tool in macOS 26.4, following the FDA clearance.
The Medical Imaging Calibration feature for Studio Display XDR has received FDA clearance and is now accessible on macOS 26.4 in the US! This enables radiologists to utilize the world’s premier professional display in general radiology. The convergence of health and technology is truly remarkable. ? pic.twitter.com/vXDIFLnHeW— Greg Joswiak (@gregjoz) April 7, 2026
While the Apple Studio Display XDR necessitates macOS 26.3.1 or higher, it is essential to note that older macOS versions may not fully exploit its advanced features. These features include high-quality Reference Modes for professional creatives, as well as the Center Stage camera and Spatial Audio speakers.
Although the starting price of $2,899 for the VESA mount adapter version or $3,299 for the stand-equipped version may seem steep, it is reasonable for professionals in creative or medical fields. Comparable high-end 12MP diagnostic displays can exceed $30,000 in cost, despite having lower resolution than Apple’s 5K display. Each type of display excels in different aspects, with diagnostic displays handling tasks such as mammography that Apple’s display cannot.
It is important to highlight that the FDA clearance for medical imaging use applies exclusively to radiologists in the United States. While Apple may seek similar clearances from other health authorities globally, the specifications for the Apple Studio Display XDR in countries like Canada, the UK, Australia, Japan, and Germany do not mention the Medical Imaging reference modes.
-

 Facebook6 months ago
Facebook6 months agoEU Takes Action Against Instagram and Facebook for Violating Illegal Content Rules
-

 Facebook6 months ago
Facebook6 months agoWarning: Facebook Creators Face Monetization Loss for Stealing and Reposting Videos
-

 Facebook4 months ago
Facebook4 months agoFacebook’s New Look: A Blend of Instagram’s Style
-

 Facebook6 months ago
Facebook6 months agoFacebook Compliance: ICE-tracking Page Removed After US Government Intervention
-

 Facebook4 months ago
Facebook4 months agoFacebook and Instagram to Reduce Personalized Ads for European Users
-
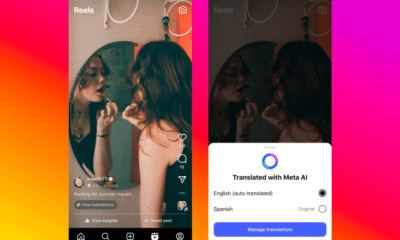
 Facebook6 months ago
Facebook6 months agoInstaDub: Meta’s AI Translation Tool for Instagram Videos
-
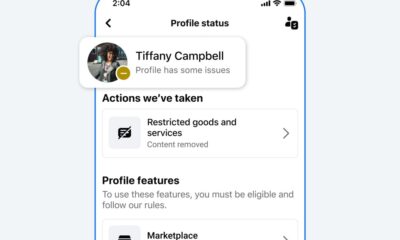
 Facebook4 months ago
Facebook4 months agoReclaim Your Account: Facebook and Instagram Launch New Hub for Account Recovery
-

 Apple6 months ago
Apple6 months agoMeta discontinues Messenger apps for Windows and macOS